ACCELERATED CARBONATION OF CALCIUM SPECIES FOR PERMANENT CARBON SEQUESTRATION IN CONCRETE
Carbon Reduction Scientists Dr Gareth Davies and Dr Viet Luan Ho have written the latest white paper ‘Accelerated Carbonation of Calcium Species for Permanent Carbon Sequestration in Concrete’. Read the white paper below.
Graphical Abstract
Introduction
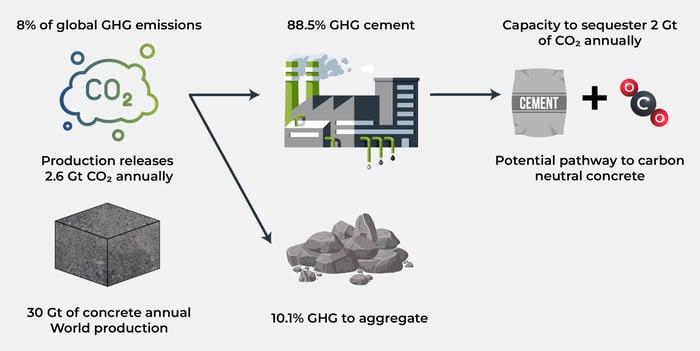
Concrete is the most widely used manmade material in the world by both weight and volume [1]. An estimated 30 Gt of concrete is produced annually, this equates to a consumption over 3.5 tonnes of concrete per person annually. The only material competing with concrete for production is fresh water, which is, as we all know, critically essential to human life. This should exemplify how reliant we are on concrete, globally, as a society. Concrete is produced from a binder material (cement), non-porous minerals varying in size (aggregate), water, and low concentrations admixtures (pigments, superplasticizers, accelerating agents, etc.,). It is estimated that concrete production alone contributed to 8% of anthropogenic GHG emissions and 3% of global energy demand. Current estimates are that 88.5% of the GHG emissions from concrete can be attributed to cement production with the majority of the remainder coming from mining of aggregates (10.1%). Approximately half of these carbon emissions are caused by the combustion of fossil fuels to provide the energy to convert raw materials into cement [2]. Whilst challenging to develop and implement, there is a clear roadmap to eliminating this half of the emission through implementation of electrical kilns and use of renewable energy. However, the remaining half of the emissions are caused by calcination of limestone which releases CO2 directly into the atmosphere. This production of CO2 is unavoidable and intrinsic to cement production. Consequently, novel, and innovative greenhouse gas mitigation strategies must be investigated, developed, and implemented quickly to mitigate against the remaining 44.25% of the GHG emissions caused by the production of concrete. Only once technologies are developed will a true plan to net zero concrete be cemented into place.
Portland cement is the most widely produced and utilised cement composition. It is made by heating limestone and clay materials in a kiln to form clinker and then grinding with gypsum [3]. Whilst several varieties of Portland cement are produced the most common is a grey cement, appropriately referred to as ordinary Portland cement. In 2011 it was reported that ~3 Gt of Portland Cement was produced globally [2]. Production of this cement generated approximately 2.6 Gt of CO2 emissions, half of which released via the previously highlighted calcination of limestone. For clarity it can be said that for every tonne of Portland cement produced there is 0.435 tonnes of CO2 emitted through calcination, which is currently unavoidable. Portland cement was developed in the early 1900s, therefore the chemistry, and chemical nomenclature are old and mostly understood within the realm of civil engineering. It is primarily composed of calcium silicates, calcium aluminates, calcium sulphate, and calcium aluminoferrites. The composition of ordinary Portland cement (CPA 32.5) is displayed in Table 1, alongside the traditional nomenclature and full chemical name.


.jpg?width=300&name=pexels-life-of-pix-2469%20(1).jpg)
-1.jpeg?width=300&name=Tunley%20Headshots%20(23)-1.jpeg)
-1.jpeg?width=300&name=Tunley%20Headshots%20(16)-1.jpeg)